- US: +1-408-610-2300
- Toll Free: +1-866-831-4085
- Become a Client
Regarding the report published, the global COVID-19 sample collection kits market size was prized by USD 12.8 billion in 2020. It is estimated to witness a 3.5% CAGR from 2021 to 2027.
A sampling of SARS-CoV-2 is the main important phase in the effectual analysis of effective infection. Counterfeit, as well as an inaccurate compilation of the sample, might cause incorrect or deceptive outcomes of the test. Accordingly, the Centers for Disease Control and Prevention (CDC) and new healthcare institutes have set up an average course of action and direction for efficient collection of samples and rationalize the use of COVID-19 sample collection kits, thus inspiring the progress. In the present situation, the lockdown has been put into practice throughout the world, the main companies are functioning ultimately to uphold the manufacture as well as development levels of swabs, along with the transportation medium.
Puritan Medical Products was expected USD 51 million deals support from the U.S. Department of Defense in August 2020. With this funding, the company put forward to increase its industrialized capability of assembles tip testing swabs. Such a type of encouraging financial support by the government is estimated to considerably strengthen the development of the COVID-19 sample collection kits market.
In 2020, the usage of sampling compilation assembly, for the diagnostics function sector, was estimated to hold the highest revenue share of the market. This is because of the incessant approvals of the investigative tests for SARS-CoV-2. A sum of 1,295 tests was accepted by the U.S. FDA, beneath EUAs; by 1st December 2020, beyond which 7 were antigen tests, 227 were molecular tests and 61 were antibody tests, for the recognition of COVID-19 disease. In addition, incessant improvement to increase the utilization of swabs for illness identification is inspiring the progress of the sector.
The research sector accounts for small access in the global COVID-19 sample collection kits market, in the conditions of making revenue. Yet, the rise in the figure of the R&D plan that organizes Covid-19 sample collection kits, to scrutinize the usefulness of a range of analytical tests, is estimated to impel the progress of the segment. In addition, wide-ranging financial support, to hold up study on SARS-CoV-2 pathogenesis, additionally increases the revenue creation, within this section.
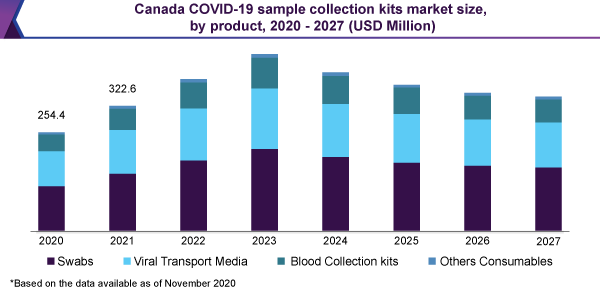
The swabs sector is projected to direct the market for COVID-19 sample collection kits. It held the 42% revenue share in 2020. The main cause is the confidence in a considerable number of accepted investigative products, make use of NP swabs. Consistent with the CDC, additional samples from the respiratory area can be collected, while the utilization of only NP swabs is not adequate. These comprise a swab from a nostril otherwise an oropharyngeal swab.
As a result of the substantial growth in the figure of hospitalizations, connected to the coronavirus, the hospitals & clinics sector was expected to hold the biggest revenue share of 93% of the COVID-19 sample collection kits market, in 2020.
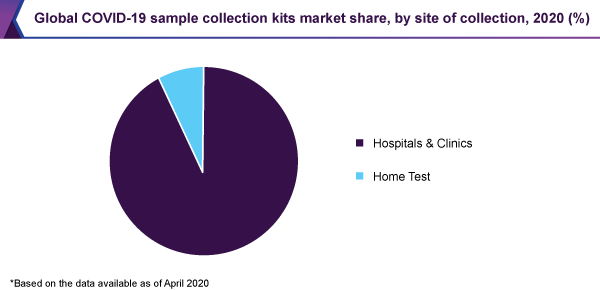
Since the figure of COVID-19 cases is globally escalating, several hospitals are increasing their testing capacity for the recognition of the SARS-CoV-2 virus. This is immediately affecting the utilization of Covid-19 sample collection kits within the hospital location, therefore consequential in the domination of this sector.
North America held a 36.1% revenue share of the global COVID-19 sample collection kits market, in 2020. It is expected to control the market. The U.S. is one of the maximum impacted nations of the world, by the maximum amount of active cases. The nation had executed more than 207 million COVID-19 tests, since 07th December 2020. This is the maximum figure of tests; some country has carried out as of now. Europe is positioning in the second place, in the conditions of revenue. The U.K. government declared, in November 2020, the proposal of fresh laboratories, to twofold the testing capability in the first part of next year.
The amenities would make possible the handing out of more than 600,000 PCR tests, every day. Conversely, the Asia Pacific section is expected to develop by the rewarding CAGR, during the period of the forecast. The government organizations are vigorously busy in sickness administration, within the state, this adds to the revenue enlargement, within the region. For example, the Indian Council of Medical Research (ICMR) introduced a movable covid-19 RT-PCR laboratory at the ICMR, in November 2020. The government proclaims its proposal to include further, such types of labs, to increase testing within the nation.
The existence of a considerable number of entrenched, medium-sized and small, companies has generated an enhancement in market rivalry. Each company is undertaking several trade plans to increase a viable upper hand in the market. Several crucial contestants of the market comprise COPAN Diagnostics, Thermo Fisher Scientific, Inc., Becton, Dickinson, and Company, and Puritan Medical Products. By way of the eruption of COVID-19, COPAN Diagnostics and Puritan Medical Products have observed, productive development in their income.
The companies are doing continuous hard work to make sure that the transportation and the circulation of the goods are done speedily as well as professionally. This contains monitoring the international logistics along with shipping arrangement, on top of taking necessary measures to conquer the capability and limitation restrictions, in the making and deliverance of COVID-19 sample collection kits.
The important participants have joined forces with local establishments, as well as suppliers, for maintaining the manufacturing circumstances and speed up the company’s expansion in the viral and swabs transportation method market. Among the continuing rush in demand, because of the COVID-19 outburst, companies are focused on their efforts on planned products to take full advantage of the product yield and delivery.
• HiMedia Laboratories
• Vitagene Inc.
• Quest Diagnostics
• Trinity Biotech
• Lucence Diagnostics Pte Ltd.
• Thermo Fisher Scientific, Inc.
• COPAN Diagnostics
• VIRCELL S.L.
• Form labs
• Danaher Corporation
• Quidel Corporation
• Hardy Diagnostics
• Laboratory Corporation of America Holdings
• Becton, Dickinson and Company
• Puritan Medical Products
|
Report Attribute |
Details |
|
The market size value in 2020 |
USD 12.8 billion |
|
The revenue forecast in 2027 |
USD 16.3 billion |
|
Growth Rate |
CAGR of 3.5% from 2021 to 2027 |
|
The base year for estimation |
2020 |
|
Historical data |
2020 (as of Nov 2020) |
|
Forecast period |
2021 - 2027 |
|
Quantitative units |
Revenue in USD Million and CAGR from 2021 to 2027 |
|
Report coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
|
Segments covered |
Product, application, site of collection, region |
|
Regional scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
|
Country scope |
U.S., Canada, Germany, U.K., France, Italy, Spain, Russia, China, India, South Korea, Australia, Japan, Brazil, Mexico, South Africa, Saudi Arabia |
|
Key companies profiled |
Puritan Medical Products; COPAN Diagnostics; Becton, Dickinson, and Company; Thermo Fisher Scientific, Inc.; Laboratory Corporation of America Holdings; Lucence Diagnostics Pte Ltd.; Hardy Diagnostics; Trinity Biotech; Quidel Corporation; Quest Diagnostics; Danaher Corporation; Vitagene Inc.; Formlabs; HiMedia Laboratories; VIRCELL S.L. |
|
Customization scope |
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope |
|
Pricing and purchase options |
Avail of customized purchase options to meet your exact research needs. |
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2020 to 2027. For this study and based on the data available as of November 2020, Million Insights has segmented the global COVID-19 sample collection kits market report based on product, application, site of collection, and region:
• Product Outlook (Revenue, USD Million, 2020 - 2027)
• Swabs
• Nasopharyngeal (NP) swabs
• Oropharyngeal (OP) swabs
• Nasal swabs
• Viral Transport Media
• Blood Collection Kits
• Other Consumables
• Application Outlook (Revenue, USD Million, 2020 - 2027)
• Diagnostics
• Research
• Site of Collection Outlook (Revenue, USD Million, 2020 - 2027)
• Hospitals & Clinics
• Home Test
• Regional Outlook (Revenue, USD Million, 2020 - 2027)
• North America
• U.S.
• Canada
• Europe
• U.K.
• Germany
• France
• Italy
• Spain
• Russia
• The Asia Pacific
• China
• India
• South Korea
• Australia
• Japan
• Latin America
• Brazil
• Mexico
• Middle East Africa (MEA)
• South Africa
• Saudi Arabia


Research Support Specialist, USA