- US: +1-408-610-2300
- Toll Free: +1-866-831-4085
- Become a Client
With reference to the report published, the global protein stability analysis market size was prized by USD 985.8 million in 2019. It is estimated to witness a 10.8% CAGR during the period of the forecast.
Protein stability analysis is a fundamental phase for the duration of medical improvement, starting from the development of active pharmaceutical ingredients (API) up to the justification of the product. Major companies are busy applying these testing methods for evaluation of the usefulness and excellence of the medicine products, beneath a variety of ecological features.
The progress of a new-fangled high throughput method, for example, Stable-seq, by the experts at The American Society for Biochemistry and Molecular Biology, Inc., that contain hereditary assortment in grouping with high throughput DNA sequencing, is expected to stimulate the development of the market. This method successfully assesses the in vivo steadiness of protein variants. This is essential for the recognition of the constituents in the proteolytic passageway, which has an effect on the protein turnover as well as connected actions.
Subcontracting has been extensively in use in the activity of the medical improvement, as a business policy, to increase efficiency in R&D actions of drug development and decrease the overheads, incurred on it. A number of contract research associations, together with 2bind GmbH and Lake Pharma Inc., are presenting investigation results of stability by means of strength, excellence, accurateness in anticipated timelines. Furthermore, falling in line with the regulatory potential, adds-on the implementation of outsourced services, by pharmaceutical manufacturing companies.
The innovators of biopharmaceuticals are at the front position of the individual reaction to the Covid-19 pandemic. A considerable amount of foremost biotech firms are in the center of a race to explore the Sars-Cov-2 genome and make ready, a possible vaccine for it. As equated to the rate of the reaction to SARS/MERs and all that, the biotech companies are scrutinizing SARs-Cov-2, at an exceptional speed and substantial sums of finances are being deposited into the R&D actions. By means of numerous contenders in the trial, the private, as well as public sectors, are expected to operate, in harmony for the estimated period, awaiting a vaccine is developed for Covid-19.
In 2019, Differential Scanning Calorimetry (DSC) retained the principal share of revenue in the protein stability analysis market. The Differential Scanning Calorimetry (DSC) is considering normal stability demonstrating method since it necessitates negligible enlargement of the assay. Furthermore, owing to its greater reproducibility and accurateness, DSC is extensively utilized in distinguishing and choosing appropriate proteins for the development of the drug and studies of ligand interaction. The additionally linked compensations comprise label-free as well as straight measurement.
Label-free Differential Scanning Fluorimetry (DSF) is a comparatively new-fangled method and shows sound development prospective, during the nearing prospect. This method has achieved huge recognition in latest years since it is appropriate for analyzing very small quantities of protein. It is simple to carry out, and it can be accomplished by means of essential and tryptophan as well as tyrosine fluorescence.
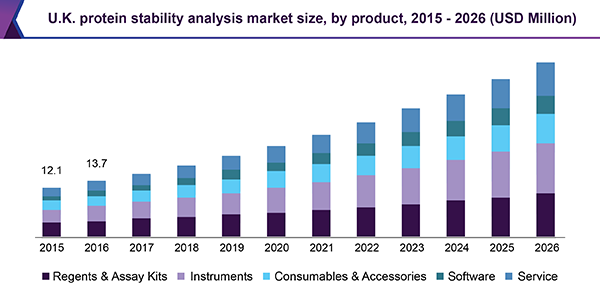
In 2019, the reagents and assay kits sector ruled the protein stability analysis market and is estimated to uphold its domination all through the period of the forecast. The execution of the reagents along with software facilitates well-organized, reasonably priced, and speedy high throughput viewing of thermal stability of the protein. It helps in the record's presentation for the recognition of ligands, which attach to the protein of concern.
The companies, like Thermo Fisher, are offering a mixture of reagents with software, which moreover permits the recognition of transformation and alterations that have an effect on the stability of the protein. Extensive applicability, at ease availability of reagents, and small price are moreover added to the major share of the sector.
Conversely, the instruments sector is anticipated to observe the highest speed of expansion for the duration of the reading. This is for the reason that these automatic methods provide, extremely receptive protein stability analysis, by means of negligible utilization of sample in a smaller structure of time. Continuous improvement in the design of the instrument, along with the integration of new-fangled features, advances efficiency throughout biopharmaceutical detection and improvement, plus create superior class, reproducible, and dependable protein denaturation information.
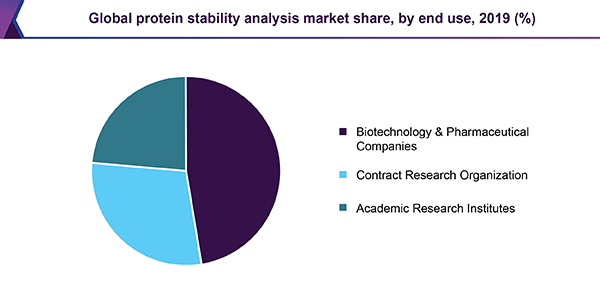
Owing to the availability of many protein therapeutics commodities in the market, intended for protein stability analysis, and a huge number of therapeutics existing in the preclinical and scientific improvement phase, biotechnology and pharmaceutical companies held the biggest share of revenue, in the protein stability analysis market. The latest authorization of recombinant DNA and monoclonal antibody technology, furthermore offer a productive opening for the improvement of novel therapeutics.
The Asia Pacific is anticipated to observe the highest percentage of development, during the nearby prospect. This is credited to the hard work, carried out by the CROs to increase their consumer support, via setting up relations with biotechnology, as well as pharmaceutical companies in Asia. Furthermore, the growing figure of clinical trials in Asia has moreover improved the acceptance of proteomics services, in the Asia Pacific.
In 2019, North America held the major share in the global protein stability analysis market. A number of initiatives carried out by the private as well as public units and the programs for financial support started by them in the proteomics industry have donated to the major share. Funding was given by the National Institute of Health (NIH), National Center for Research Resources, US HUPO, and additional government entities, speed up the actions of proteomic research within this region.
For Example, the Disruptive Proteomics Technologies (DPT) Working Group of NIH has concentrated on the recognition of gaps and openings in methodologies and technologies, on the subject of protein analysis. This methodology of NIH facilitates significant progression in protein stability analysis, through an extensive variety of biomedical research fields.
Crucial companies are taking on considerable lead to strengthen their existence in the market and retain a viable upper hand in the place. Besides, contestants of the market are caught up in the contract policy, joint venture forms, development of the product, and geographic expansion, in untouched regions.
• Applied Photo physics Ltd.
• Unchained Labs
• Agilent Technologies, Inc.
• HORIBA, Ltd.
• NanoTemper
• Enzo Biochem, Inc.
• Waters Corporation
• SETARAM Instrumentation
• Malvern Panalytical Ltd.
• GE Healthcare
• PerkinElmer Inc.
• Thermo Fisher Scientific, Inc.
|
Report Attribute |
Details |
|
The market size value in 2020 |
USD 1.10 billion |
|
The revenue forecast in 2026 |
USD 2.0 billion |
|
Growth Rate |
CAGR of 10.8% from 2020 to 2026 |
|
The base year for estimation |
2019 |
|
Historical data |
2015 - 2018 |
|
Forecast period |
2020 - 2026 |
|
Quantitative units |
Revenue in USD Million and CAGR from 2020 to 2026 |
|
Report coverage |
Revenue forecast; company share; competitive landscape; growth factors and trends |
|
Segments covered |
Product, technique, end-use, region |
|
Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
|
Country scope |
U.S.; Canada; Germany; U.K.; China; Japan; Brazil; South Africa |
|
Key companies profiled |
Thermo Fisher Scientific, Inc; Enzo Biochem, Inc.; PerkinElmer Inc.; NanoTemper; GE Healthcare; HORIBA, Ltd.; Malvern Panalytical Ltd.; Agilent Technologies, Inc.; SETARAM Instrumentation; Unchained Labs; Waters Corporation; Applied Photophysics Ltd. |
|
Customization scope |
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country; regional & segment scope. |
|
Pricing and purchase options |
Avail of customized purchase options to meet your exact research needs. |
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2015 to 2026. For the purpose of this study, Million Insights has segmented the global protein stability analysis market report on the basis of product, technique, end-use, and region:
• Product Outlook (Revenue, USD Million, 2015 - 2026)
• Reagents and Assay Kits
• Instruments
• Consumables and Accessories
• Software
• Service
• Technique Outlook (Revenue, USD Million, 2015 - 2026)
• Chromatography
• Spectroscopy
• Differential Scanning Calorimetry (DSC)
• Differential Scanning Fluorimetry (DSF)
• Dynamic Light Scattering (DLS)
• Others
• End-use Outlook (Revenue, USD Million, 2015 - 2026)
• Pharmaceutical & Biotechnology Companies
• Contract Research Organizations
• Academic Research Institutes
• Regional Outlook (Revenue, USD Million, 2015 - 2026)
• North America
• The U.S.
• Canada
• Europe
• Germany
• The U.K.
• The Asia Pacific
• China
• Japan
• Latin America
• Brazil
• Middle East and Africa (MEA)
• South Africa


Research Support Specialist, USA